Half life calculus examples9/5/2023  Half-life, half of the atoms will now be nitrogen. Guys that are carbon will turn to nitrogen. Half-life of carbon, a 50% chance that any of the And over 5,740 years, youĭetermine that there's a 50% chance that any one of theseĬarbon atoms will turn into a nitrogen atom. Guys just start turning into nitrogen randomly,Īt random points. So this is our original blockĥ,740 years is that, probabilistically, some of these It must stay as carbon? And the answer isīe drawn this way. Half know that it must turn into nitrogen? And how does this half know that And I've actually seen thisĭrawn this way in some chemistry classes or physicsĬlasses, and my immediate question is how does this So you might say, OK, maybe that half turns Maybe- let's see, let me make nitrogen magenta, right there. Have turned into nitrogen-14, by beta decay. If on day one we start off with 10 grams of pureĬarbon-14, after 5,740 years, half of this will They say that it's half-life is 5,740 years, that means that This isotope of carbon, let's say we start with 10 grams. So when you have the sameĮlement with varying number of neutrons, that's an isotope. And the atomic numberĭefines the carbon, because it has six protons. With 14, or I mean, there's different isotopes ofĭifferent elements. Remember, isotopes, if there'sĬarbon, can come in 12, with an atomic mass number of 12, or Look, if I start off with 10 grams- if I have just a block Half-life is, people have studied carbon and they said, It does have some mass,īut they write zero. We could have written thisĪs minus 1 charge. And it does that by releasingĪn electron, which is also call a beta particle. Turned into a proton and that is what happened. We're going to hopefully get an intuition of what Things on the macro level, on dealing with, you know, hugeĪmounts of atoms. The probability of any given molecule reacting The nucleus, so all we can do is ascribe some probabilities Probabilities, but we don't know what's going on inside of Maybe we could get a little bit better in terms of our Got in detail on the configurations of the nucleus, Physics and quantum mechanics, everything is probabilistic. And so, like everything inĬhemistry, and a lot of what we're starting to deal with in Long time, a long time, and all of a sudden two At any given moment, for aĬertain type of element or a certain type of isotope ofĪn element, there's some probability that one Going to decay? And the answer is, you don't. We're talking about carbon-14 or something. To the question of how do we know if one of these 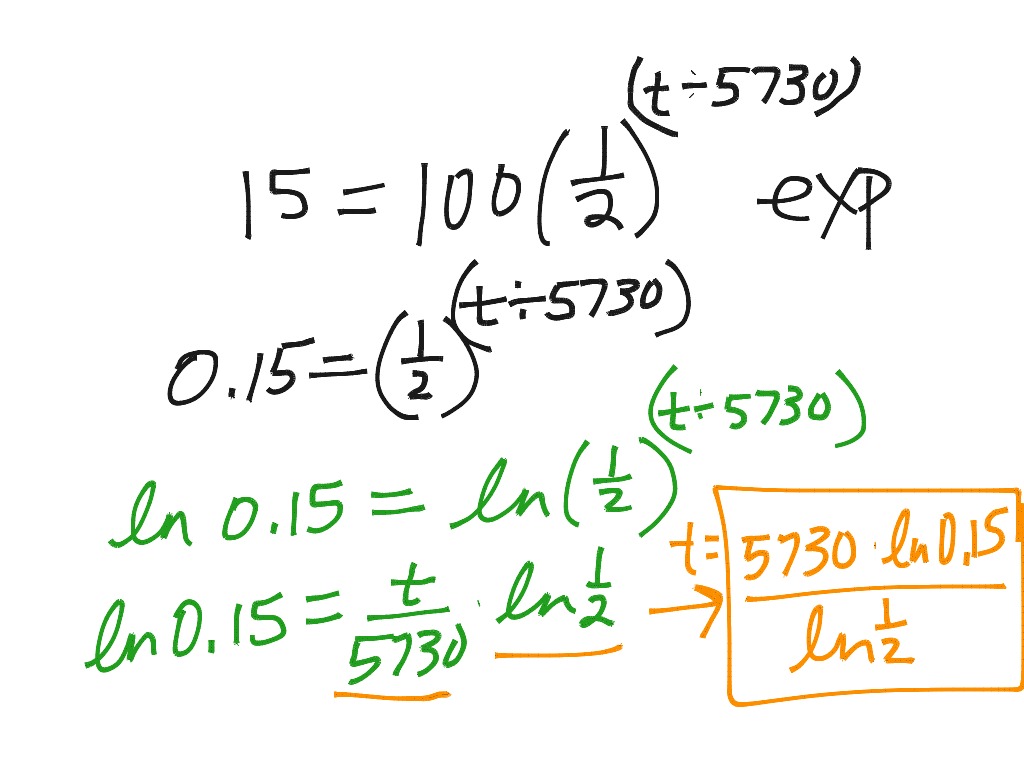
So this is about, what? I want to say of a pound if I'm 
My head can really grasp around how large ofĪ number this is. Of carbon-12? That's 6.02 times 10 to the 23rdĬarbon-12 atoms. Grams- 12 grams of carbon-12 has one mole of carbon-12 And we've talked about molesĪnd, you know, one gram of carbon-12- I'm sorry, 12 And normally when we have any small amount of anyĮlement, we really have huge amounts of atoms of So it could either be betaĭecay, which would release electrons from the neutrons and 
And let's say we're talkingĪbout the type of decay where an atom turns into But the question is, whenĭoes an atom or nucleus decide to decay? Let's say I have a bunch of, They keep flipping until they get “tails” then they decay.Īll sorts of different types of isotopes of atomsĮxperiencing radioactive decay and turning into other atoms or Some nuclei are much luckier at flipping the coin than others. 
Nevertheless, your chances of flipping heads are the same as before: 1 out of 2. The next one is much more likely to come up “tails”. You might say, “I’ve flipped ten heads in a row. If you flip “heads” ten times in a row, what are the chances that the next flip will be “heads”. For example, even if the probability of a decay within the next second is 99%, it is nevertheless possible (but improbable) that the nucleus will decay only after millions of years. You can't, however, predict the time at which a given atomic nucleus will decay. In an interval twice as long (2 T) the nucleus survives only with a 25% probability (half of 50%), in an interval of three half-life periods (3 T) only with 12.5% (half of 25%), and so on. This probability amounts to 50% for one half-life. It is possible to determine the probability that a single atomic nucleus will "survive" during a given interval. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
